Polycythemia vera (PV) and essential thrombocythemia (ET) are myeloproliferative neoplasms (MPNs) characterized by phenotype defining somatic mutations of JAK2, cMPL, and CALR genes. These mutations activate the JAK-STAT signaling pathway, leading to augmented cellular proliferation. A major cause of morbidity and mortality in PV and ET is thrombosis. Current therapy options include hydroxyurea (HU), interferon-α (IFN-α), and JAK2 inhibitors. IFN-α treatment normalizes blood cell counts, and reduces thrombotic complications in PV and ET and has been associated with a decrease in JAK2V617F variant allele frequency (VAF), indicating its selective targeting of JAK2V617F-mutated cells. IFN-α treatment also rescues the normal dormant HSCs, resulting in conversion of clonal to polyclonal myelopoiesis (Tashi, Leukemia, 2018). Ropeginterferon alfa (ropeg-IFNα)-2b is mono-pegylated IFNα-2b with greater tolerability and an extended half-life compared to Peg-IFNα-2α and is FDA approved for PV therapy.
We recently discovered a patient with a novel germline JAK2R715T mutation, which causes erythrocytosis with PV-like features (Kim, ASH, 2022, Song under review in Blood). This patient had poor tolerance to HU and a JAK2 inhibitor therapy; although the latter normalized her blood cell counts, it was poorly tolerated. She has been on ropeg-IFNα-2b since Mar. 2023. Given that ropeg-IFNα-2b's known function is to reduce mutated cells (Verger, Blood Cancer J. 2018) and all her hematopoietic cells were bearing a JAK2 gain-of-function germline mutation, we did not expect a complete hematological response, but her blood counts completely normalized. This observation led us to hypothesize that ropeg-IFNα-2b induces remission by reduction of JAK2 activity.
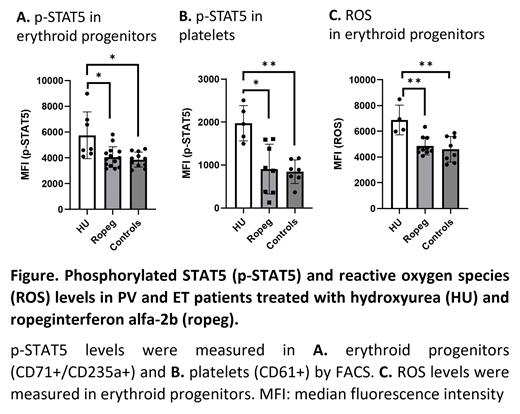
To measure JAK2 activity, we quantitated phosphorylated STAT5 (p-STAT5) levels in her erythroid progenitors (CD71 +/CD235a +) and observed a 20% reduction in these levels after ropeg-IFNα-2b treatment. This prompted us to investigate PV and ET patients with the typical JAK2V617F mutation treated with ropeg-IFNα-2b. We measured p-STAT5 in erythroid progenitors and platelets (CD61 +) of 8 PV and 5 ET patients treated with ropeg-IFNα-2b and 1 PV and 6 ET patients treated with HU. Levels of p-STAT5 were higher in patients treated with HU compared to those treated with Ropeg-IFNα-2b (Figure A and B). In healthy controls, levels of p-STAT5 were lower than those in HU treated patients but comparable to those in ropeg-IFNα-2b treated patients (Figure A and B). p-STAT5 levels showed a positive correlation with platelet counts (r=0.5235, p=0.0397) and neutrophil counts (r=0.6088, p=0.0235). p-STAT5 levels were then measured before and after ropeg-IFNα-2b treatment in 1 ET patient and showed a 20% reduction, which coincided with a decrease in platelet count from 746 x 10 9/L to 207 x 10 9/L.
Peg-IFN-2α downregulates upregulated oxidative stress genes and upregulates downregulated antioxidative defense genes in MPNs (Skov, PLOS One, 2022), suggesting its potential role in reducing oxidative stress.We measured reactive oxygen species (ROS) in erythroid progenitors and found that ROS levels were significantly higher in PV and ET patients treated with HU compared to those treated with ropeg-IFNα-2b and to healthy controls (Figure C). JAK2V617F mutated cells accumulate more ROS compared to unmutated cells (Marty, Leukemia, 2013). We found a positive correlation between ROS levels and p-STAT5 levels (r=0.4778, p=0.0449), suggesting an interaction between JAK2 activity and oxidative stress in PV and ET.
Our study demonstrated the impact of ropeg-IFNα-2b treatment on JAK2 activity in PV and ET. Ropeg-IFNα-2b effectively reduced JAK2 activity, leading to suppression of dysregulated cell proliferation, and decreased ROS accumulation. To minimize the influence of confounding factors that could independently affect JAK2 activity, we are now comparing samples from the same patients before and after ropeg-IFNα-2b treatment. These results suggest a novel molecular benefit of ropeg-IFNα-2b therapy in PV and ET.
Disclosures
No relevant conflicts of interest to declare.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal